 
First, calculate the amount of ingredient in 250mL of a 25 solution. If the concentration of the original stock solution was 100 µg/µL, the concentration in Tube 4 would beġ00 µg/µL × #1/160000# = 6. Basic dilutions Serial dilutions Dilutions for concentrated waters Triturations. The dilution factor after four dilutions is Repeat the process until you have four tubes. Then transfer 0.2 mL from Tube 2 to 3.8 mL of diluent in Tube 3 and mix. You would transfer 0.2 mL from Tube 1 to 3.8 mL of diluent in Tube 2 and mix. So you multiply each successive dilution by the dilution factor. Remember that serial dilutions are always made by taking a set quantity of the initial dilution and adding it successively to tubes with the same volume. If you did the above dilution four times, what would be the final dilution factor? What is the dilution factor if you add 0.2 mL of a stock solution to 3.8 mL of diluent? Diluting a solution resulting from a previous dilution by adding the same amount of solvent is known as a serial dilution. 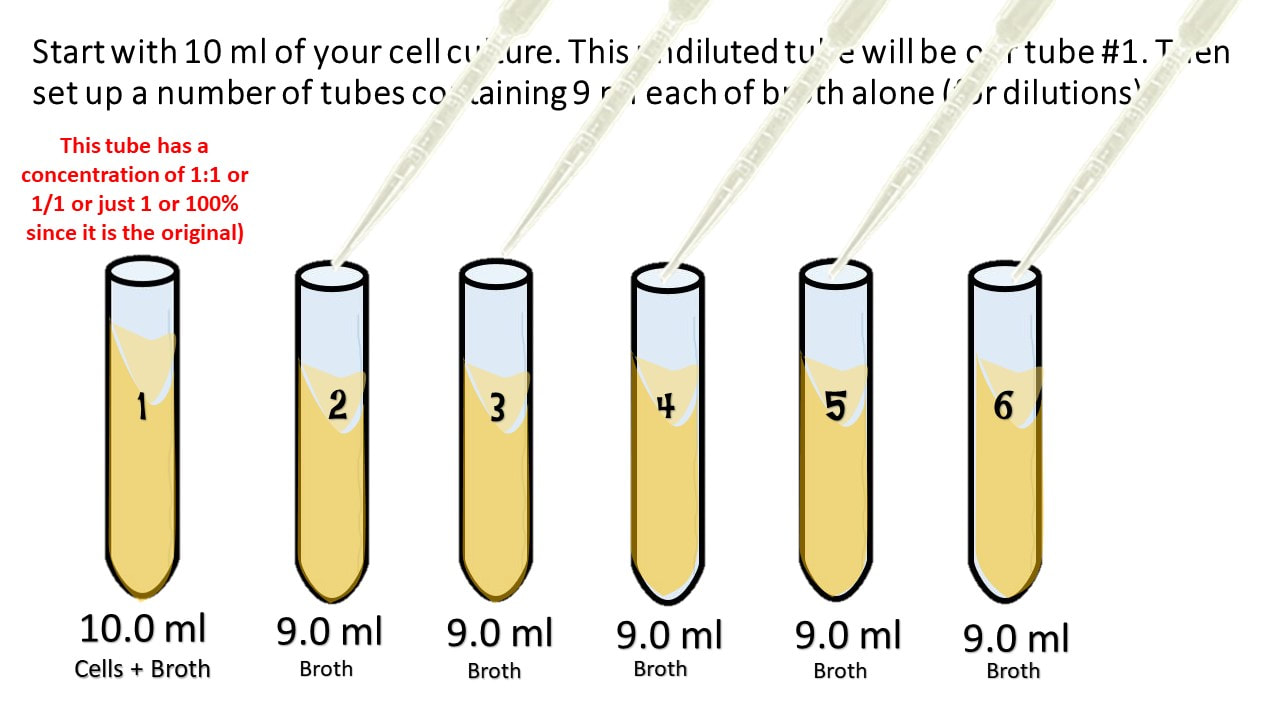
The dilution factor or the dilution is the initial volume divided by the final volume.įor example, if you add a 1 mL sample to 9 mL of diluent to get 10 mL of solution, In serial dilutions, you multiply the dilution factors for each step. You do serial dilutions on a water sample, and plate the dilutions on TSA plates.A serial dilution is any dilution in which the concentration decreases by the same factor in each successive step. Explanation: A serial dilution is any dilution in which the concentration decreases by the same factor in each successive step. Given the information below, fill in the number of colonies you would expect on each of the plates.ĥ. Serial Dilution Calculator How do you calculate serial dilutions You multiply the original concentration by the dilution factors for each dilution. You do a series of dilutions as shown below, and you plate 1.0 ml of each dilution. The concentration of antibody required: /. The formula used is: Concentration (stock) × Volume (stock) Concentration (dilute) × Volume (dilute) The stock concentration of your antibody: /. How many colonies will you see at each dilution?Ĥ. The antibody dilution tool calculates the volume of stock antibody and buffer needed in order to achieve a solution of the desired volume and concentration. You do serial dilutions to achieve a 1/10,000 dilution, and then plate 0.1 ml of each of these dilutions. Used to determine how much liquid is needed to resuspend a number of moles to a. This calculator is useful for diluting primers and DNA oligos. The antibody dilution tool calculates the volume of stock antibody and buffer needed in order to achieve a solution of the desired. The tool below can be used to create a protocol for preparing a serial dilution from a stock solution. For instance, creating a two-fold dilution with a starting concentration of 10 M yields the following concentrations: 10 M, 5 M, 2.5 M, 1.25 M, etc. This calculator is useful for diluting DNA samples. A serial dilution is a sequence of dilutions created using the same dilution factor. Dilute solution to a desired concentration (mass/vol). You have a bacterial culture that you know has 650,000 bacteria/ml. Molecular Biology Dilution & Solution Calculators. You want to make a 1/100,000 fold dilution, but the smallest volume you can measure is 1.0 ml, and the tubes available to you only hold 10 ml. You have received a sample from a sewage treatment plant, and have been asked to determine how many CFUs/ml are in this sample. How many CFUs/ml were in the original urine sample?Ģ. You make ten-fold dilutions of this sample as shown below, and then plate 0.5 ml (500 µl) of the last dilution on a TSA plate. A ten-fold serial dilution, which can also be called a 1:10 dilution, or a series with dilution factor of 10. You have a urine sample from a patient that you suspect has a urinary tract infection. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
